If you own a healthcare facility or provide a service to one, you know how important it is to be HIPAA compliant. HIPAA has a long list of requirements, and documentation is an important one. The struggle with HIPAA documentation is often confusing. The legal speak often leads non-compliance folks down the rabbit hole.
But worry not, we have made it easier for you. In this article, you will learn what HIPAA document requirements are, why it is important, and how to maintain HIPAA documents.
What is HIPAA documentation?
HIPAA documentation is a combination of policies, processes, and tracking per the requirements mandated by the Security and Privacy Rules of HIPAA.
This is a necessary process that enables you to gain visible demonstrable evidence (VDE). The documentation can be achieved using electronic means such as HIPAA compliant software or maintained on non electronic formats like paper.
HIPAA documentation requirements arise from three primary rules, each governing a distinct area of compliance:
- Privacy Rule: Governs the use and disclosure of protected health information (PHI), individual rights, and organizational policies around data access and sharing
- Security Rule: Covers administrative, technical, and physical safeguards required to protect electronic PHI (ePHI)
- Breach Notification Rule: Mandates documentation of security incidents, breach assessments, and reporting obligations to affected individuals and regulators
Understanding which rule each documentation requirement falls under helps organizations prioritize their efforts and ensures no area of compliance is overlooked.
Why is HIPAA documentation important?
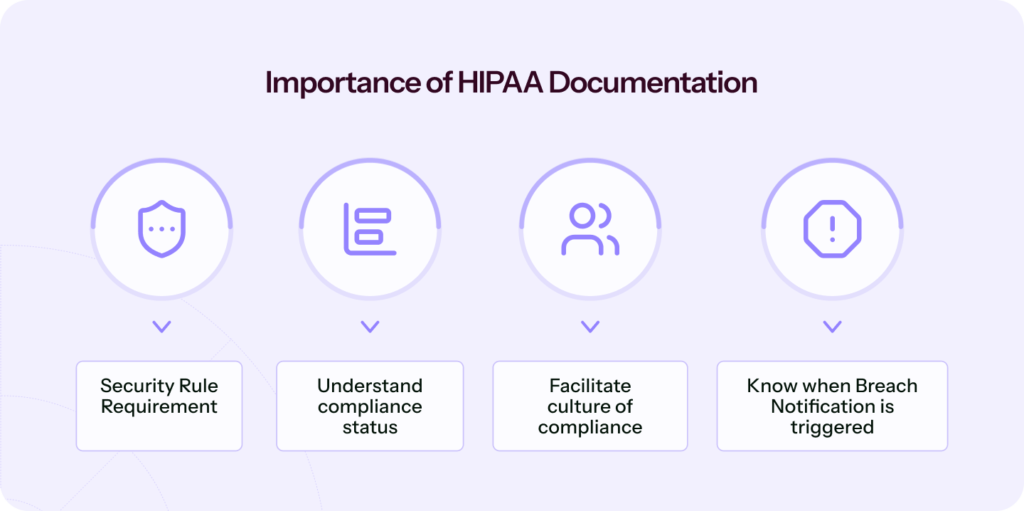
HIPAA documentation is a requirement of the security rule. If you are a business associate or covered entity, you should not consider this requirement a burden to avoid legal issues.
It helps compliance officers understand how compliant you are and what actions are yet to be implemented. It shows how your facility functions and helps with maintaining records and thus helps to demonstrate transparency.
HIPAA documentation facilitates a culture of compliance. It is necessary to determine when there has been a violation of the privacy rule to take corrective action against the responsible employee and determine if the breach notification has been triggered.
What are the HIPAA documentation requirements?
HIPAA documentation requirements span three rules, each with distinct obligations for covered entities (CEs) and business associates (BAs). Before diving in, two foundational concepts apply across all requirements:
Required vs addressable specifications
Security Rule implementation specifications are classified as either required or addressable. Required specifications must be implemented without exception. Addressable specifications are not optional if an organization chooses not to implement one, it must conduct and document a formal risk analysis explaining why it is not reasonable or appropriate for its environment, and what equivalent alternative measure has been put in place. If a standard adopted in 164.308, 164.310, 164.312, 164.314, or 164.316 includes implementation specifications, CEs and BAs must document why it is not necessary to implement the concerned specification.
Mandatory risk analysis
A documented risk analysis is a non-negotiable baseline requirement under the Security Rule. It must identify:
- Threats and vulnerabilities to ePHI
- The likelihood and potential impact of each threat
- Current controls in place and their effectiveness
Beyond the initial analysis, organizations must maintain a risk management plan and conduct ongoing reviews as systems and threats evolve. CEs and BAs must also review or modify security measures needed to adequately protect ePHI and update relevant documents as per 164.316(b)(2)(iii).
Privacy rule documentation
The Privacy Rule governs how PHI is used, disclosed, and protected. It also requires organizations to clearly define where PHI lives and who is responsible for it. CEs and BAs must document:
Designated Record Sets and PHI Management:
- What constitutes a designated record set within the organization
- Where PHI is stored and who maintains it
- The method used by an expert to determine that certain health information does not qualify as PHI
- Covered entities must document designation in written or electronic format and designate parts of health components in keeping with paragraph (c)
Access and Minimum Necessary Policies:
- Minimum necessary policies governing who can access PHI and under what circumstances
- Role-based access controls and information limitation procedures
- Written designation of health components and authorization logs for every patient
Individual Rights and Authorizations:
- Signed authorizations stored and retained as per 164.530(j)
- Restrictions on PHI use or disclosure as per 164.530(j)
- Notice requirements as stated in 164.530(j)
- Titles of individuals who receive and process amendment requests, retained as required documents
- The data required in accounting for PHI disclosure, written accounting provided to individuals under section 164.528, and the title of people who receive and process requests for accounting
- A written acknowledgment of receipt of privacy notices as per (c)(2)(i) — or documented efforts to obtain one if acknowledgment was not received
- Research documentation obtained from researchers in cases involving deceased individuals
- Plan sponsors must adequately document the difference between them and group health plans
Complaints, Sanctions, and Training:
- All complaints received must be documented by CEs
- Sanctions against employees who fail to comply with [HIPAA privacy](HIPAA privacy) policies
- Administrative requirements including staff training records
- Any agreement or assurance documented in a Business Associate Agreement (BAA)
“Sprinto integrates with everything that we use, and collects evidence automatically. Centralizing evidence in one place is critical for us, so it’s nice that Sprinto does this out-of-the-box.” – Deepak Balasubramanyam, CTO, Rocketlane
Want compliance that runs itself? See it in action. Book a demo
Security rule documentation
The Security Rule governs the protection of ePHI through administrative, technical, and physical safeguards. As per 164.306, CEs and BAs must maintain policies and processes related to this subpart in written or electronic form. CEs and BAs must document:
Administrative Safeguards:
- Risk analysis and risk management policies, reviewed and updated regularly
- Security incidents and their outcomes
Workforce Lifecycle Documentation:
- Workforce clearance procedures for new hires and contractors
- Access provisioning and deprovisioning records tied to role changes and terminations
- Sanction policy documentation outlining consequences for policy violations
- Training logs evidencing completion of required security awareness programs
Technical Safeguards:
- Unique user ID assignments and audit controls
- Activity review logs and automatic logoff procedures
- Policies governing ePHI transmission security
Physical Safeguards:
- Facility access controls and workstation use policies
- Policies or processes to document corrections or updates to physical security components of a practice
- Device and media control procedures including disposal and reuse policies
Contingency Planning:
- Data backup plan documenting how ePHI is backed up and how frequently
- Disaster recovery plan outlining procedures to restore lost data
- Emergency mode operations plan to maintain critical business processes during a crisis
- Testing and revision procedures to ensure contingency plans remain effective and up to date
Where an implementation specification is deemed not necessary, CEs and BAs must document the rationale and any alternative measures implemented in its place.
Breach notification rule documentation
The Breach Notification Rule mandates documentation of incidents and reporting obligations. CEs and BAs must document:
- Security incidents and their outcomes, including the scope and nature of the breach
- A log of breaches affecting fewer than 500 individuals
- Notifications provided to affected individuals, regulators, and media where applicable
- If a law official orally requests that a notice or posting would impede a criminal investigation, the CE or BA must document that statement
Retention requirements
All HIPAA documentation must be retained for a minimum of six years from the date of creation or the date it last went into effect, whichever is later, as per 164.306. This applies to policies, procedures, activity logs, and any written records of assessments or decisions.
Learn more about HIPAA with this video:
How to manage your organization’s HIPAA documents
The tracking method should allow you to track and even produce VDE to all HIPAA requirements. In order to achieve this, you must have:
- A comprehensive understanding of all requirements applicable to your facility.
- You must be able to produce VDE for each of these requirements.
- If you are unable to produce VDE for some reason, there must a strategy to achieve the same.
- You should be able to track every requirement at a granular level.
As previously outlined, both hard copies or electronic means are acceptable to maintain the records. However, an electronic format is preferable due to ease of use and sustainability.
Conclusion
Let’s get one thing straight. HIPAA certification and documentation requirements are long and not easy to maintain. A small miss can land you in legal trouble or interrupt your workflow. Doing it manually is tedious, time-consuming, and error-prone – it can quickly mutate and become a mess.
There is a better, smarter, easier, and faster way to get this done – the Sprinto way. How does it work, you ask? Sprinto has all the requirements built into it. Sprinto captures logs, documents every granular detail you may ever require, tracks unauthorized actions, and automates all your documentation requirements. It also gives you the visibility required to track your compliance posture and identity areas where you are currently non-compliant and predict future non-compliance scenarios.
“Sprinto offered the flexibility we needed. With Sprinto we could see how we could cross-use controls between frameworks to drive HIPAA and ISO27001:2013 in parallel. And the teams’ prompt response and clear communication sealed the deal!” – Sriram Subramanian, Consultant Director of IT Compliance, CareStack
Let’s make compliance easier. Talk to our experts about your needs.
FAQs
How do you document patient information?
You can document patient information using an electronic format like a HIPAA compliant cloud solution or non-electronic format like paper.
What should you document as per HIPAA?
As per the privacy and security rule of HIPAA covered entities and business associates must document policies, processes, and procedures related to patient health information.
What documents does HIPAA protect?
HIPAA protects sensitive patient health information or protected health information (PHI) documented in electronic or non-electronic format. There are 18 identifiers that qualify as PHI.
Author
Anwita
Anwita is a cybersecurity enthusiast and veteran blogger all rolled into one. Her love for everything cybersecurity started her journey into the world compliance. With multiple certifications on cybersecurity under her belt, she aims to simplify complex security related topics for all audiences. She loves to read nonfiction, listen to progressive rock, and watches sitcoms on the weekends.
Reviewer
Rachna Dutta
Rachna Dutta is an Information Security Consultant at Sprinto and a certified ISO 27001 Lead Auditor with expertise across a broad spectrum of global and India-specific compliance frameworks, including PCI DSS, SOC 2, HIPAA, FedRAMP, HITRUST, NIST CSF, and RBI regulatory requirements.Explore more
research & insights curated to help you earn a seat at the table.